Developmental Hematopoiesis
Bryder Group
Our Research
Our research focuses on how hematopoietic stem cells (HSCs) and their immediate offspring are regulated in situations such as infections, stress, aging and leukemia. Although HSCs are associated with a fairly clear functional definition regarding their developmental and self-renewal potential, all HSCs do not behave the same.
We have developed and applied FACS-based high-resolution cell fractionation procedures to map the differentiation processes from HSCs. This has allowed for direct assessments of the molecular properties that associate with distinct developmental stages of hematopoiesis, and represents a powerful experimental paradigm when for instance applied to the study of experimental transgenic models. Aberrant regulation of blood cell formation underlies a wide range of hematological disorders, including leukemia. By studying murine transgenic leukemia models developed in our laboratory, we are currently exploring the relationships between hematopoietic progenitor cell identities and the development and progression of acute leukemia.
The production of distinct blood cell types can be selectively adapted to meet an individual’s acute needs, such as for instance the rapid and enhanced generation of innate immune cells following infection. This highlights the flexibility and adaptability of the control mechanisms that govern hematopoiesis. In addition, HSCs are unique in that they can also produce new HSCs via self-renewal; a basic but key requirement to ensure life-long blood cell production. A sub line of our research deals with how proliferation dynamics, at defined cellular stages, associate with these processes.
The blood cell system is associated with many age-related changes. These include amongst others a decreased resistance and response to infections, and an increased incidence of pathologies, including leukemia. Due to the hierarchical structure of hematopoiesis, we have suggested that many of these changes can be associated with an altered HSC function with age. We have established an influence of both compromised genomic maintenance (nuclear and mitochondrial), and alterations in external growth control to these processes, which also have direct relevance for the stability and potential reversibility of the aging stem cell state.
Aims
A broader interest of our laboratory is to understand the molecular regulation of HSCs both from the perspectives of self-renewal and differentiation, and how these features become altered upon disease-causing or disease-associated perturbations. Our goal is to understand, at cellular and molecular levels, what controls HSCs and the fate of other immature blood cells. Traditionally, such evaluations have been made using transplantation-based models, but recent and ongoing studies, including from our laboratory, have shown that HSC function in this setting is fundamentally different from normal HSC function.
Impact
We believe such enhanced knowledge can be used both preventively and therapeutically, by being able to expand the use of stem cell transplantation as a therapy, but also in the development of stem cell-based therapies that do not involve direct transplantation.
List of Team Bryder's publications
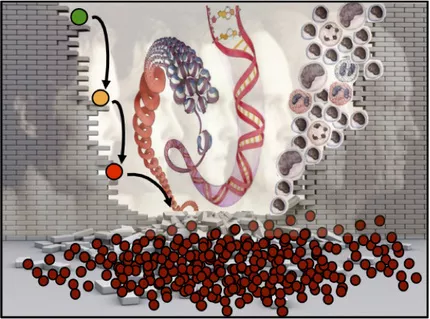
A diverse range of functional distinct effector cells (right) constitutes the building bricks of the blood cell system. The mature blood cells are all generated from immature hematopoietic stem cells (HSCs, represented in green), which reside at the top of the hierarchy.
Team
(name linked to profile in Lund University research portal)
David Bryder
Principal Investigator, David [dot] Bryder [at] med [dot] lu [dot] se (David[dot]Bryder[at]med[dot]lu[dot]se)
Qinyu Zhang
Postdoc, Qinyu [dot] Zhang [at] med [dot] lu [dot] se (Qinyu[dot]Zhang[at]med[dot]lu[dot]se)
Anna Konturek
PhD student, Anna [dot] Konturek-Ciesla [at] med [dot] lu [dot] se (Anna[dot]Konturek-Ciesla[at]med[dot]lu[dot]se)
Jinyuan Wang
Student, jinyuan [dot] wang [at] med [dot] lu [dot] se (jinyuan[dot]wang[at]med[dot]lu[dot]se)
Yahya Hama
Student, ya6847ha-s [at] student [dot] lu [dot] se (ya6847ha-s[at]student[dot]lu[dot]se)
David Bryder
Principal Investigator
PhD, Professor
Division of Molecular Hematology
Department of Laboratory Medicine
Lund Stem Cell Center
BMC B12, Lund University
221 84 Lund, Sweden
Phone: + 46 46 222 39 51
Mail: David [dot] Bryder [at] med [dot] lu [dot] se (David[dot]Bryder[at]med[dot]lu[dot]se)
Profile in Lund University research portal