Vector production charges
Lentiviral and Retroviral Vector production charges for StemTherapy researchers:
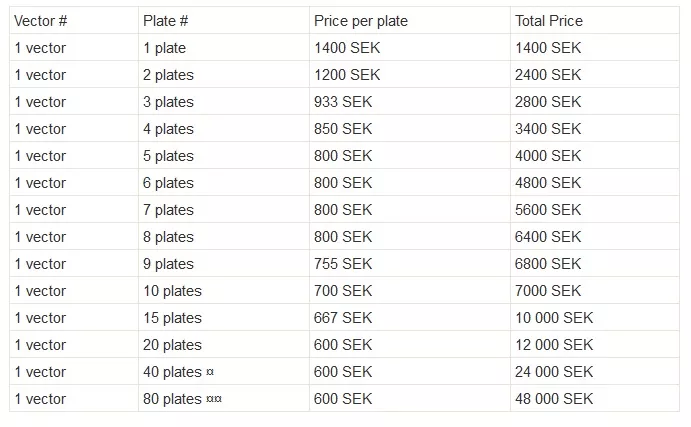
The Full-Service Vector production prices below include vector production (using the user-provided transfer vector plasmid), concentration of the vector prep via ultracentrifugation, and titration by FACS. If titration by Q-PCR is required, this can be arranged for a 600 SEK surcharge per order plus 300 SEK per vector.
Notes:
- Lentiviral and retroviral vector production charges are identical.
- Lentiviral vector preps and retroviral vectors preps require different amounts of transfer vector plasmid per plate. Thus provide 7ug/40ug DNA per plate for Lenti and 10ug per plate for Retro.
- There is no longer a discount for vector production orders with multiple different transfer vectors. For example, a request for single plate vector production of each of 5 unique transfer vectors, the cost is 5 x 1400 SEK= 7000.
- If Q-PCR based titration is required an extra charge of 600 SEK per order plus 300 SEK per vector is levied.
Maximum order size:
- The maximum number of plates that will be produced for a user is 20 plates per week. I.e. if 40 plates ordered the vector production will be split into 2 rounds performed on available weeks. 80 plates will be split into 4 production rounds to be performed on available weeks.
Saving money:
If ultracentrifugation or titration are not required by user, the total cost wil be reduced by 30% and 20%, respectively. For example 50% price reduction if vector is produced with neither ultracentrifugation nor titration. (Note: Orders without ultracentrifugation and titration, the maximum plate order per user is increased to 40 plates; please discuss with Vector Research Engineer beata [dot] lindqvist [at] med [dot] lu [dot] se (Beata Lindqvist)).
Additional costs for external users of the vector core production facilities:
The cost for our services for academic customers in Sweden outside of the Malmö/Lund University area is as in table above plus 60%.
The costs for our services for international academic institutions is as in table above plus 125%.
The cost for our services for external commercial customers is as above plus 200%
QuickTest production charges for StemTherapy researchers:
The purpose of the quick test is to provide a quick and less expensive evaluation of novel vectors that allows user to evaluate gene expression and function. It is able to be offered to reserachers since it is significantly less work for the vector unit staff as production is performed without ultracentrifugation concentration of vector. Production is performed only on 10cm dishes whereby visual confirmation of vector functionality is provided (if fluorescent marker gene present). The transfected cells and supernatant is provided to the user for analysis by the user (the dish is provided without processng the supernatant or cellular material).
Single plate/well transfection of novel vector
Price includes a visual report on marker gene expression followig transfection and delivery of the plate (no processing of supernatant or cellular material:
450 SEK/plate
Quicktest functional test of vector
A biological function confirmation of the non-concentrated vector prep is avaialble where HT1080 cells are transduced (single well only 500uL of virus) and marker gene assessed after 3-4 days post infection by FACS. The transduced cells maybe provided to the user upon request.
300 SEK/vector