Laboratory of Stem Cells and Restorative Neurology
Kokaia group
Aim
By linking together basic and clinical research, we aim to clarify cellular mechanisms of regeneration following damage to the brain and develop new therapeutic strategies to restore function in this organ (primarily in stroke and Parkinson’s disease) by transplantation of stem cells or reprogrammed cells and optimization of endogenous repair mechanisms.
Our Research
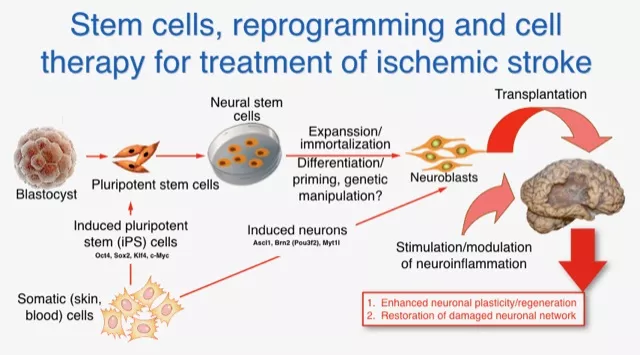
Available experience, intellectual potential and manpower, technology platforms, transgenic animals, methodological and clinical “know-how”, and close interaction between clinicians and basic scientists provide added value, strongly supporting the feasibility of our work. We are transplanting different types of neuroblasts generated either from human skin cell-derived induced pluripotent stem (iPS) cells or via direct conversion of skin cells to neurons (iN cells) (Fig.1).
Reprogramming of somatic (e.g., skin or blood) cells is an emerging technology which gives the possibility to develop any cell type avoiding the ethical concerns with the use of human embryonic stem (ES) cells. In addition, using iPS and iN cell technology patient-specific cells can be generated for transplantation, avoiding the need for immunosuppression and risk for rejection. We are currently generating neurons by iPS and iN cell technology aiming to produce cortical neurons and after intracerebral transplantation or direct in vivo reprogramming of non-neuonal cells restore damaged neuronal network.
We also determine how inflammation associated with ischemic brain damage influences the plasticity of the brain and how the beneficial effects of innate and adaptive immune system could be promoted to improve functional recovery. We are currently performing clinical study to characterize the phenotype and gene expression profile of peripheral blood monocytes in patients with stroke.
Impact
List of Team Kokaia's publications
Our laboratory for the first time combines transplantation of stem cells, stimulation of endogenous neurogenesis and modulation of inflammatory responses in order to develop clinically effective cell replacement therapies for human neurodegenerative diseases. Such diseases represent leading causes of disability, and with the growing aging population, their economical and societal burden will be further increased. If our objectives are achieved, this work will have major therapeutic implications for patients with neurodegenerative disorders such as stroke.
Team
(name linked to profile in Lund University research portal)
Zaal Kokaia
Principal Investigator, Zaal [dot] Kokaia [at] med [dot] lu [dot] se (Zaal[dot]Kokaia[at]med[dot]lu[dot]se)
Olle Lindvall
Senior Professor, Olle [dot] Lindvall [at] med [dot] lu [dot] se (Olle[dot]Lindvall[at]med[dot]lu[dot]se)
Sibel Aktay
Postdoc, Sibel [dot] Aktay [at] med [dot] lu [dot] se (Sibel[dot]Aktay[at]med[dot]lu[dot]se)
Zhengyu Fang
Postdoc, Zhengyu [dot] Fang [at] med [dot] lu [dot] se (Zhengyu[dot]Fang[at]med[dot]lu[dot]se)
Sara Palma Tortosa
Postdoc, Sara [dot] Palma_Tortosa [at] med [dot] lu [dot] se (Sara[dot]Palma_Tortosa[at]med[dot]lu[dot]se)
Juliane Tampe
PhD student, Juliane [dot] Tampe [at] med [dot] lu [dot] se (Juliane[dot]Tampe[at]med[dot]lu[dot]se)
Natalia Avaliani
Research Engineer, Natalia [dot] Avaliani [at] med [dot] lu [dot] se (Natalia[dot]Avaliani[at]med[dot]lu[dot]se)
Linda Jansson
Research Engineer, Linda_I [dot] Jansson [at] med [dot] lu [dot] se (Linda_I[dot]Jansson[at]med[dot]lu[dot]se)
Emanuela Monni
Research Engineer, Emanuela [dot] Monni [at] med [dot] lu [dot] se (Emanuela[dot]Monni[at]med[dot]lu[dot]se)
Katarina Turesson
Research Administrator, Katarina [dot] Turesson [at] med [dot] lu [dot] se (Katarina[dot]Turesson[at]med[dot]lu[dot]se)
Zaal Kokaia
Professor Zaal Kokaia
Principal Investigator
Phone: +46 46 2220276
Mobile: 0705-365917
Email: Zaal [dot] Kokaia [at] med [dot] lu [dot] se (Zaal[dot]Kokaia[at]med[dot]lu[dot]se)