Nearly half of all cancerous brain tumors are glioblastomas, an aggressive type of cancer. This type of tumor grows rapidly and can spread throughout the brain making it difficult to treat - with about 40% of patients surviving the first-year post-diagnosis. Treatments, which typically include surgery followed by radiation and chemotherapy, mainly work to prolong, and improve a patient’s quality of life by easing symptoms and slowing cancer growth.
To improve treatment options and outcomes for patients, a team of researchers at Lund Stem Cell Center, based at Lund University and Skåne University Hospital in Lund, are exploring whether a potential treatment can be developed by reprogramming cancer cells into cells that, as far as possible, function normally.
“We had the idea to use a reprogramming approach on these aggressive, malignant brain tumor cells. Reprogramming is a technique that can activate certain genes that can redirect one cell type into another. Here, we wanted to push the cancer cells towards something less harsh, less aggressive,” explained Johan Bengzon, Professor of Neurosurgery.
Reprogramming glioblastoma tumor cells into astrocytes
Glioblastomas are thought to arise from the cancerous transformation of glial cells, the brain’s support cells. Current research indicates that astrocytes - star-shaped glial cells that actively participate in brain signaling and are necessary for the healthy function of neurons and the brain as a whole - are often co-opted to contribute to glioblastoma progression, meaning they can become cancerous and support tumor growth.
“By taking advantage of the similarities between neural developmental processes and cancer formation, we can attempt to correct the tumor and possibly eliminate the malignant nature of the tumor cells,” stated Francesco Trovato, Postdoctoral Reseacher.
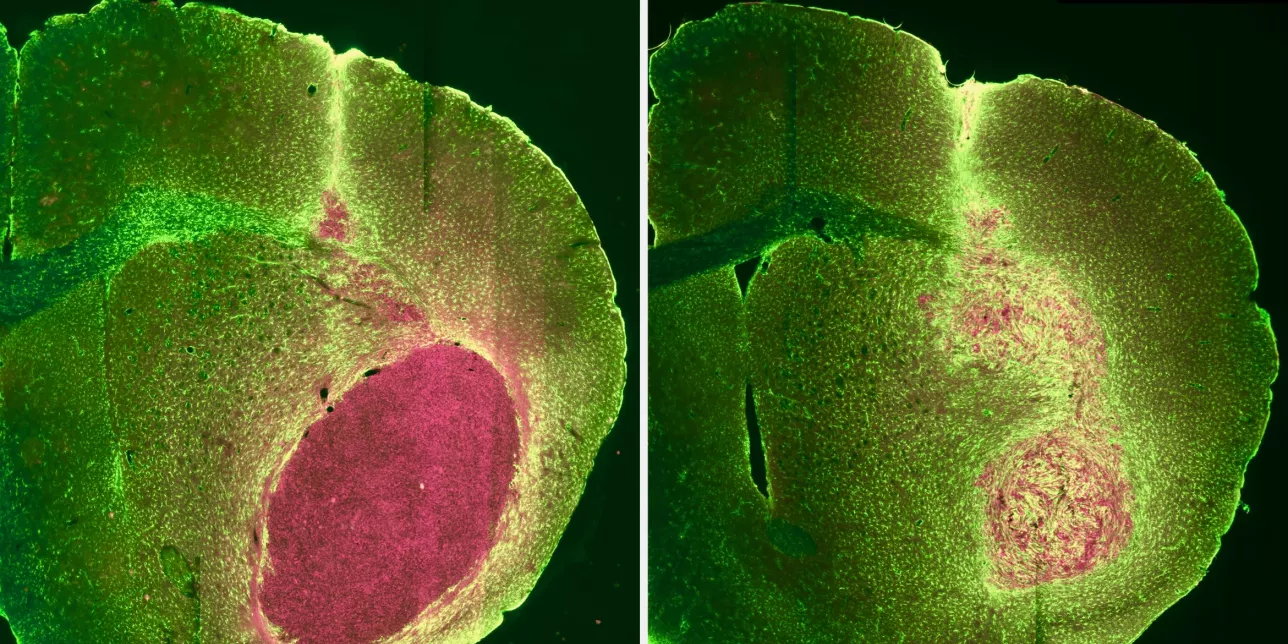
In a proof-of-concept study now published in Molecular Cancer Therapeutics, Francesco Trovato, a postdoctoral researcher in the labs of Henrik Ahlenius and Johan Bengzon, reprogrammed human glioblastoma cells and glioblastoma stem cells, triggering them to develop into mature, astrocyte-like cells.
“The reprogrammed cells acquired astrocyte-like structures and functions. The cells expressed typical glial characteristics, showed a marked decrease in their ability to multiply, and most importantly, exhibited a reduced ability to produce tumors,” described Henrik Ahlenius, Researcher and Principal Investigator.
The results of this new study build on earlier work from the Ahlenius laboratory, which in 2018 presented a method to generate astrocytes from human pluripotent stem cells and, earlier this year, described the efficient reprogramming of human fibroblasts, or skin cells, into astrocytes.
“We hope this method of turning cancer cells into mature, astrocyte-like cells will pave the way for new therapeutic approaches to be added to the treatment toolbox for glioblastoma,” noted Henrik Ahlenius.
Advancing the treatment toolbox for glioblastoma
Based on the findings from this study, the research team has received additional funding to bring their discoveries into the clinic as a potential new gene therapy for patients with glioblastoma and possibly other types of brain tumors.
With funding from the Erling Persson Foundation (4 million SEK) and Barncancerfonden (1 million SEK), Henrik Ahlenius and Johan Bengzon plan to advance their research to the next stage. They will do this by replicating their method in a pure in vivo setting and reprogramming the tumor cells inside the brains of their animal models. The reprogramming method will also be developed further using human tumor tissue removed during glioblastoma surgery.
“Using human glioblastoma tissue collected during surgery, we will repeat this process using organoids and slice cultures as glioblastoma tissue models. This is the most accurate representation of the processes occurring in human patients that we can recreate in the lab,” revealed Johan Bengzon.
Postdoctoral researcher Francesco Trovato will also continue to explore the use of reprogramming both to dampen glioblastoma tumor expansion and progression and as a model for childhood and adult tumors, thanks to a 3 million SEK postdoctoral fellowship grant he received earlier this spring from the Swedish Society for Medical Research.
“Over the next three years, we intend to implement techniques that are not only possible in a lab setting but can also be applied clinically. We plan to combine reprogramming with genetic engineering to generate specific, genetically tailored models of adult and childhood brain tumors which can be used to evaluate therapies or as a model system to answer certain basic biological questions,” concluded Francesco Trovato.