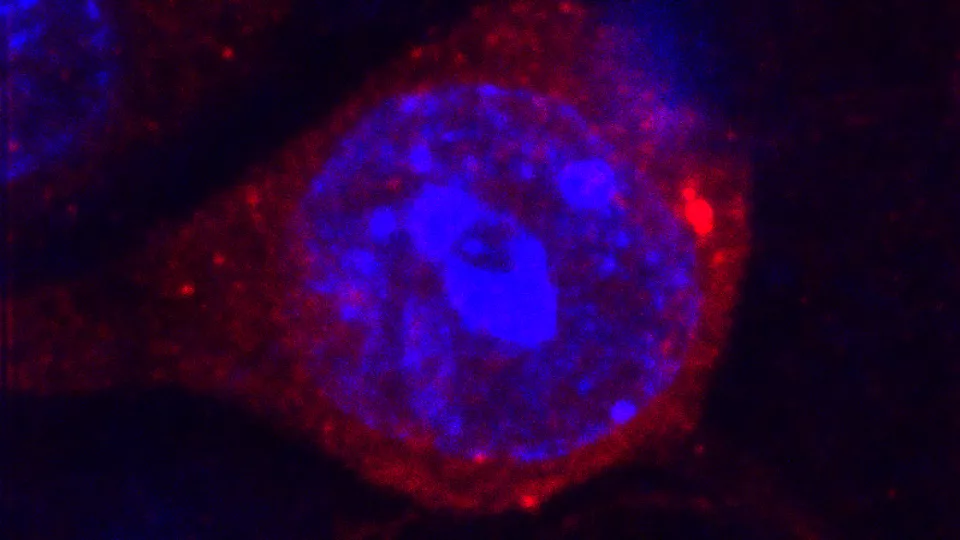
Around 10% of the human genome is composed of DNA sequences originating from viruses that infected our ancestors throughout the millions of years of our evolution. Under normal conditions these ancient viral elements - known as endogenous retroviruses or ERVs for short - are inactivated by a complex process known as epigenetic silencing.
The aberrant activation of ERVs has been described in various types of human disease, including neurological disorders such as Schizophrenia, Multiple Sclerosis and Parkinson’s disease. Despite this, cause and effect studies directly linking ERVs to the development of brain disorders have been lacking - until now.
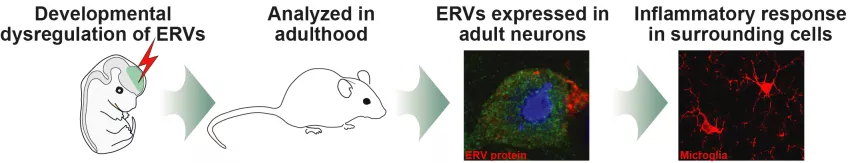
In a recent study, the Molecular Neurogenetics group - led by Prof. Johan Jakobsson - show dysregulation of ERVs during development can lead to both inflammation in the brain and gene expression patterns associated with various neurological disorders later in life.
“By using CRISPR-Cas9 gene editing technology we could generate several mouse models in which the silencing of specific ERVs is inactivated, either in the developing or in the adult brain,” explains Marie Jönsson, PhD, senior scientist, and first author of the study recently published in the EMBO journal.
“We observed that the active epigenetic silencing of ERVs is crucial during brain development, as these ancient viral elements were switched on and maintained their expression in the adult brain,” explains Marie.
Strikingly, the effects of ERV activation were not limited to neurons in which the epigenetic silencing was lost, but were also seen in adjacent cell types in which the epigenetic silencing was unaffected.
“When activated, ERVs produced not only genetic material but also proteins which agitated adjacent cells such as microglia - immune cells acting as a first line of defense for the brain and involved in processes such as inflammation,” continues Marie.
The research team hypothesize that this phenomenon contributes to the inflammatory response often seen in psychiatric or neurodegenerative disorders.
“These exciting findings demonstrate how vital the epigenetic control of ERVs are during brain development,” concludes Marie “They also highlight that events affecting this control during brain development could play an important role in the development of neurological disorders much later in life.”
Publication in the EMBO Journal: Activation of endogenous retroviruses during brain development causes an inflammatory response.